Health authorities caution that weight-loss injections may lead to unexpected vision loss, prompting the introduction of new safety warnings.
Health authorities issue urgent warnings about weight-loss injections, revealing a rare risk of vision loss linked to semaglutide treatments amid rising safety concerns.

UK regulators just made a scary tweak to semaglutide’s official warnings, and it’s one of those updates that instantly makes everyone pause. The new product info flags a link to NAION, a rare condition that can trigger sudden, painless vision loss.
The twist is that semaglutide sits at the center of two major conversations right now: diabetes control and weight-loss injections. People are taking it under brand names like Rybelsus and Ozempic, and others are using Wegovy for weight goals, so the warning doesn’t live in some far-off corner. It lands right where “it works” is already a daily headline, and now the eye-safety angle is getting louder.
And once those warnings start spreading, the real question becomes: how many people will notice the symptoms in time?
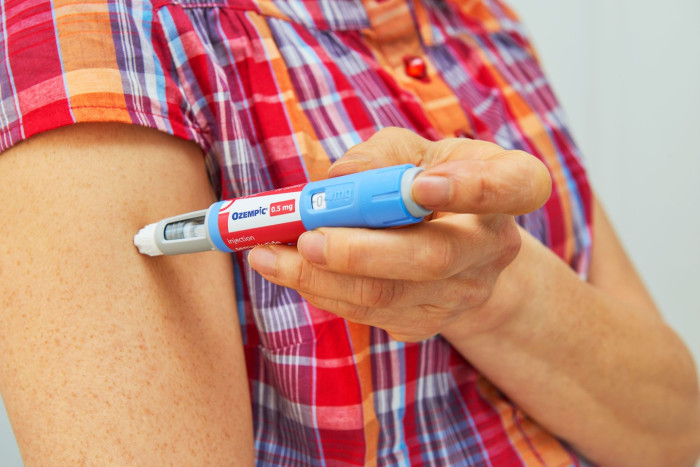
UK Revises Semaglutide Warnings for Eye Health Risks
The UK's health regulatory authority has taken decisive action by revising the product information for semaglutide treatments, now highlighting the association with non-arteritic anterior ischemic optic neuropathy (NAION). This decision stems from a growing body of evidence that suggests a correlation between the medication and this rare eye condition, which can lead to sudden vision loss.
As healthcare providers begin to reassess their prescribing practices, patients are urged to remain vigilant and report any unusual visual symptoms promptly. The unfolding situation emphasizes the need for ongoing research and monitoring to ensure patient safety while navigating the complexities of obesity and diabetes management.
That’s when the “rare but real” NAION warning started showing up alongside the usual semaglutide buzz, like it was always part of the plan until now.
Semaglutide: Effective for Diabetes and Weight Loss
It is marketed under various brand names, including Rybelsus and Ozempic for diabetes management, and Wegovy specifically for weight loss. The benefits of semaglutide are well-documented, with numerous studies highlighting its effectiveness in helping patients achieve significant weight loss and improved glycemic control.
However, a study published in 2024 brought to light a concerning complication: cases of vision loss associated with the use of semaglutide. This revelation has prompted health officials to take precautionary measures to ensure patient safety.
Semaglutide Linked to Risk of 'Eye-Stroke'
Experts have indicated that semaglutide may lead to a condition colloquially referred to as an 'eye-stroke.' This occurs when the arteries that supply blood to the eyes become blocked, resulting in decreased blood flow to the optic nerve. The implications of this condition can be severe, as it may lead to sudden and painless vision loss, typically affecting one eye.
Although the occurrence of NAION is extremely rare, with only three cases reported in the UK since semaglutide was first authorized for use in 2018, the potential risk has raised alarms among healthcare professionals and regulatory bodies. In response to these findings, health officials in the UK have updated their guidance to include warnings about NAION for patients using Wegovy.
Sudden Vision Loss from NAION: Key Insights
NAION is characterized by a sudden reduction in blood flow to the front part of the optic nerve, which can result in abrupt vision loss. The condition is particularly concerning because it can occur without warning and does not present with pain, making it difficult for patients to recognize the severity of their symptoms until it is too late.
Alison Cave, the Chief Safety Officer at the Medicines and Healthcare products Regulatory Agency (MHRA), emphasized the importance of patient safety in her statement regarding the updated guidance."
It got messier after a 2024 report surfaced, tying semaglutide to sudden vision loss cases and pushing regulators to revise the product information.
It also brings to mind Trump’s baffling reaction to a reporter asking his Valentine’s Day plans with Melania.
Monitoring NAION Risks with Semaglutide Use
Cave further highlighted that while the potential risk of NAION associated with semaglutide is exceedingly small, it is crucial for both patients and healthcare professionals to be vigilant about the symptoms that may indicate the onset of this condition. Patients who are prescribed semaglutide are now advised to seek immediate medical attention if they experience any sudden changes in their eyesight.
This includes any abrupt impairment of vision or a rapid deterioration of eyesight in one or both eyes. The MHRA's updated guidance serves as a critical reminder for patients to remain aware of their health and to act promptly if they notice any concerning symptoms.
Even with only three UK cases reported since 2018, the phrase “eye-stroke” still hits hard, especially when the medicine is being used for everyday weight and diabetes routines.
Semaglutide Prescriptions Now Warn of Blindness Risk
In light of these developments, semaglutide prescriptions will now include a warning regarding the risk of blindness, underscoring the seriousness of the potential side effects associated with this medication. The manufacturer of semaglutide, Novo Nordisk, has responded to these concerns by reaffirming its commitment to patient safety.
In a statement, the company expressed that it takes reports of adverse events very seriously and collaborates closely with regulatory authorities worldwide to monitor the safety profile of its products. Novo Nordisk has also updated the Summary of Product Characteristics (SmPC) and patient leaflets for Wegovy, Ozempic, and Rybelsus to include information about NAION.
Semaglutide Update: Rare NAION Adverse Effect Identified
This update is reflected in the section detailing special warnings and precautions for use, as well as in the section outlining adverse drug reactions. The company has categorized NAION as a very rare adverse effect, indicating that it may affect up to 1 in 10,000 individuals taking semaglutide.
Despite the updated warnings, Novo Nordisk maintains that the overall benefit-risk profile of semaglutide remains favorable. The company has stated that, based on the totality of evidence, there is no reasonable possibility of a causal relationship between semaglutide and NAION.
Semaglutide: Weighing Benefits Against NAION Risks
This assertion is crucial for patients who rely on semaglutide for managing their diabetes and weight, as it suggests that the benefits of the medication may still outweigh the risks for the majority of users. The emergence of NAION as a potential side effect of semaglutide highlights the importance of ongoing research and monitoring in the field of pharmaceuticals.
As new medications are developed and introduced to the market, it is essential for regulatory bodies to remain vigilant in assessing their safety and efficacy. The case of semaglutide serves as a reminder of the complexities involved in medication management, particularly for individuals with chronic conditions such as diabetes and obesity.
Now patients are being told to stay alert for unusual visual symptoms, because one sudden change in sight is not something you can wait out.
Empowering Patients Through Transparent Medication Education
Furthermore, this situation raises broader questions about the role of healthcare providers in educating patients about the risks and benefits of their medications. It underscores the necessity for transparent communication between patients and healthcare professionals, ensuring that individuals are fully informed about the potential side effects of their treatments.
This is particularly important in the context of medications like semaglutide, which have garnered significant attention for their effectiveness in weight management and diabetes control. In conclusion, the recent updates regarding semaglutide and the associated risk of NAION serve as a critical reminder of the importance of patient safety in the realm of pharmaceuticals.
While the risk of this rare side effect is low, the implications for those affected can be severe.
Engaging Stakeholders for Optimal Patient Outcomes
As research continues to evolve, it is imperative that all stakeholders remain engaged in the conversation to promote the best outcomes for patients.
The injection may help the scale, but nobody wants it to cost them their vision.
Wait, did you see what Trump said about uncertainty in his afterlife during that peace board meeting?